Clinical Presentation and Diagnosis
Rates of EBV+ PTLD after SOT and HCT
SOT
The highest rate of EBV+ PTLD in the SOT setting occurs in the first year after transplant1,2
A second peak occurs 7 to 10 years after transplant2
After SOT median time to PTLD onset 4-5.3 years1
4-5.3
Years(1)
due to taking lifelong immunosuppressives1
HCT
EBV+ PTLD usually occurs within 12 months1
Only 4% of PTLD cases develop after 12 months1
After HCT median time to PTLD onset 2-4 months3
2-4
Months(3)
Cases occurring >5 years after HCT are extremely rare due to discontinued use of immunosuppressives1
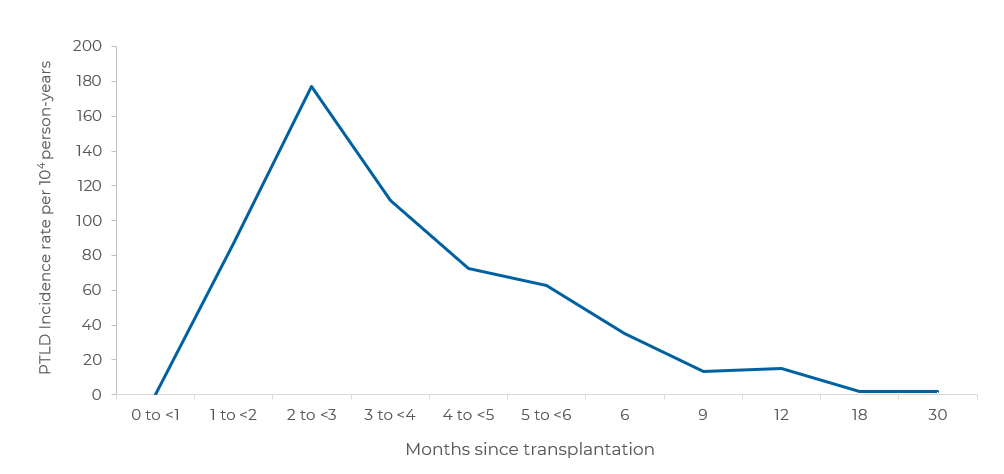
Rate of EBV+ PTLD after HCT
In a multicenter, retrospective study of 127 patients with PTLD after allogeneic HCT, incidence rates for PTLD peaked at 2 to 3 months after transplant, and then declined sharply with increasing time since transplantation3
Time to PTLD Diagnosis after SOT, by Organ Transplanted
A single-center, retrospective study found that heart transplantation had a numerically longer time from transplant to PTLD diagnosis versus other SOTs4
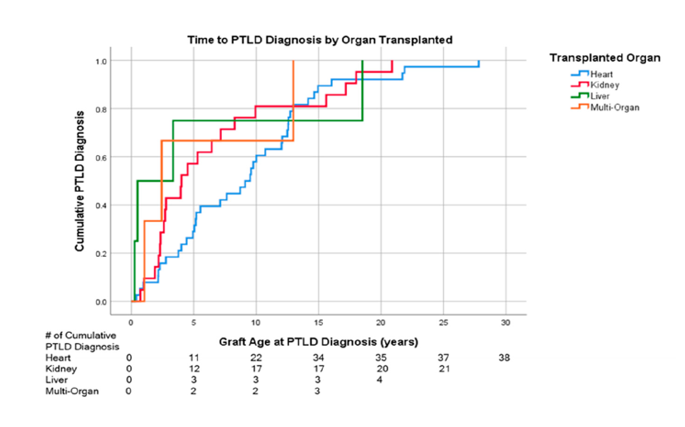
Median time to diagnosis of PTLD by organ type*:
- Liver: 0.49 years (n=4; 95% CI: 0–3.5)
- Multi-organ: 2.4 years (n=3; 95% CI: 0.21–4.6)
- Kidney: 4 years (n=21; 95% CI: 1.4–6.6)
- Heart: 9.1 years (n=38, 95% CI: 6.2–12.0)
Adapted from Lau E, et al. Cancers (Basel). 2021. https://creativecommons.org/licenses/by/4.0/.
*Out of 66 patients identified with a histologically-confirmed diagnosis of PTLD, 50 were EBV+, 10 were unknown, and 6 were EBV-negative.
Key risk factors for EBV+ PTLD after SOT and HCT
SOT2,6
Early PTLD risk factors (<12 months after transplant)
- Primary EBV infection derived from the donor
- Younger age (children)
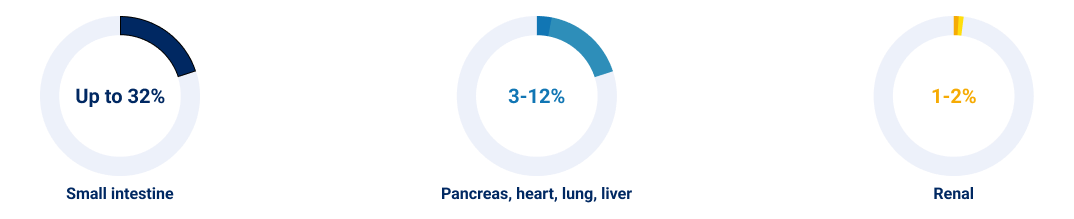
- Type of organ transplanted : Incidence of EBV+ PTLD in recipients based on organ type2,7
- Treatment with anti-lymphocyte antibodies
Late PTLD risk factors (>12 months after transplant)
- Duration of immunosuppressive treatment
- Type of organ transplanted
- Older age (adults)
HCT5,6
Pre-transplant risk factors
- Degree of T-cell reduction and impairment
- EBV sero-mismatch
- Stem cell source (i.e., cord blood transplantation)
- HLA mismatch
Post-transplant risk factors
- Severe acute/chronic GvHD
- High/rising EBV viral load (DNAemia)
- Treatment with mesenchymal stem cells

Symptoms and presenting features
The symptoms of EBV+ PTLD vary as the patient progresses from asymptomatic EBV infection to fulminant PTLD and may include1–7,10:
- Malaise and lethargy
- Fever or night sweats
- Sore throat
- Enlargement or swelling of the lymph nodes
- Weight loss
- Chronic sinus congestion
- Abdominal pain
- Nausea and vomiting
- Anorexia
- Gastrointestinal symptoms (e.g., bleeding, bowel perforation)
- End-organ disease (e.g., hepatitis)
As EBV+ PTLD progresses, it can involve any organ, such as the gastrointestinal tract, bone marrow, liver, spleen, lung, kidney, and CNS1,10
In patients with EBV+ PTLD, fever and lymphadenopathy are associated with rapidly progressive multi-organ failure and death5
Monitoring recommendations
In addition to vigilant monitoring for symptoms, surveillance following transplant should include testing for EBV viral load (DNAemia) to inform a pre-emptive treatment strategy against PTLD6,7
Consider assessing EBV viral load for SOT and HCT patients at increased risk of PTLD:2,6
- SOT recipients
- EBV-seronegative recipients receiving seropositive donor organs and EBV seronegative paediatric patients
- EBV-seropositive children <1 year of age and intestinal transplant recipients
- Recipients of high-risk allogeneic HCT
Recommendations for monitoring EBV viral load
|
|
Testing Method |
Sample Type |
Timing |
|---|---|---|---|
|
SOT2,8 |
qPCR |
Whole blood, plasma, lymphocytes |
Suggested weekly to biweekly over the high-risk period (first-year) |
|
HCT5 |
qPCR |
Whole blood, plasma, serum |
Begin within the first month |
Diagnostic and evaluation recommendations for EBV+ PTLD
For patients in whom progression to EBV+ PTLD is suspected:
The gold standard to confirm PTLD diagnosis is histological examination and immunophenotyping of tumor tissue, preferably through excisional biopsy or resection of suspected sites of disease; if not possible, core needle biopsy is an alternative5,7,12
Additional diagnostic confirmation can involve non-invasive methods, including quantification of EBV viral load (DNA-emia) and PET-CT/CT imaging5,7,12,13 and can be considered for patients unable to undergo tissue biopsy5
Diagnostic workup Includes:5,12,13
- Physical exam and evaluation of performance status
- History of immunosuppressive regimen
- PET-CT/CT imaging
- Tissue biopsy with ISH/IHC staining for viral antigens and/or flow cytometry
- EBV viral load (DNAemia) and serology assessment
- Laboratory assessments: complete blood count with differential and metabolic panel (e.g., liver enzymes, renal function)
- Bone marrow evaluation in select cases
- Endoscopy in cases of gastro-intestinal symptoms
- Echocardiography where appropriate
- Fertility-preserving treatment for eligible patients
Staging of EBV+ PTLD

Classification of PTLD according to WHO-HAEM5
The 2022 revision of the World Health Organization (WHO) Classification of Haematolymphoid Tumours (WHO-HAEM5) incorporated significant changes to the categorisation of lymphoproliferative disorders associated with immunodeficiencies to introduce an overarching framework14
Relationship between PTLDs as defined in WHO-HAEM4 and WHO-HAEM514
Click to show new classification:
WHO-HAEM4
WHO-HAEM5
Non-destructive PTLD
Hyperplasias—histologic subtypes are:
- Follicular hyperplasia
- Mononucleosis-like hyperplasia
- Plasma cell hyperplasia
- KSH/HHV8 MCD
EBV+ in most cases 15
Polymorphic PTLD
Polymorphic LPDs
The majority is EBV+; onset can be early or late after transplant13
Monomorphic PTLD
Lymphoma
The majority of early-onset disease is EBV+ while late-onset monomorphic DLBCL-like subtype is often EBV−15
CHL-PTLD
(Classic Hodgkin) Lymphoma
CHL-PTLD is an uncommon subtype16 and the majority is EBV+15,16
Nomenclature in the immune deficiency/dysregulation setting according to WHO-HAEM5
WHO-HAEM5 has additionally introduced a standardised nomenclature to encompass the various settings of immune dysfunction14
Three-part nomenclature for lymphoid proliferations and lymphomas14
| Histological diagnosis | Viral association | Immune deficiency/dysregulation setting |
|---|---|---|
|
|
|
Adapted from Alaggio R, et al., et al. Leukemia. 2022. https://creativecommons.org/licenses/by/4.0/. Table title abridged.
References
- Fujimoto A, Suzuki R. Cancers (Basel). 2020;12(2):328.
- Allen UD, Preiksaitis JK. Clin Transplant. 2019;33(9):e13652.
- Landgren O, et al. Blood. 2009;113(20):4992-5001.
- Lau E. Cancers (Basel). 2021;13(4):899.
- Styczynski J, et al. Haematologica. 2016;101(7):803–811.
- Yamada M, et al. J Pediatric Infect Dis Soc. 2024;13(Suppl 1):S31–S38.
- Nijland ML, et al. Transplant Direct. 2015;2(1):e48.
- Clerico M, et al. J Clin Med. 2022;11(24):7542.
- Lindsay J, et al. Curr Opin Infect Dis. 2021;34(6):635–645.
- Dierickx D, Habermann TM. N Engl J Med. 2018;378(6):549–562.
- Sureda A, et al. The EBMT Handbook: Haematopoietic Stem Cell Transplantation and Cellular Therapies. 8th edition. Cham (CH): Springer; 2024.
- Shah N, et al. Br J Haematol. 2021;193(4):727–740.
- Samant H, et al. Posttransplant Lymphoproliferative Disorders. StatPearls Publishing; 2023.
- Alaggio R, et al. Leukemia. 2022;36(7):1720–1748.
- Atallah-Yunes SA, et al. Br J Haematol. 2023;201(3):383–395.
- Rosenberg AS, et al. Am J Hematol. 2016; 91(6): 560–565.
Abbreviations
- CHL Classic Hodgkin Lymphoma
- CNS central nervous system
- CT computed tomography
- DLBCL diffuse large B-cell lymphoma
- EBV(+) Epstein–Barr virus (positive)
- FDG 18F-fluorodeoxyglucose
- GvHD graft-versus-host disease
- HCT haematopoietic cell transplantation
- HHV8 human herpesvirus-8
- HLA human leukocyte antigen
- IHC immunohistochemistry
- ISH in situ hybridisation
- KSH(V) Kaposi sarcoma-associated herpesvirus
- LPDs lymphoproliferative disorders
- MCD multicentric Castleman disease
- PET positron emission tomography
- PTLD post-transplant lymphoproliferative disorder
- qPCR quantitative real-time polymerase chain reaction
- SOT solid organ transplant
- WHO World Health Organization
- WHO-HAEM World Health Organization Classification of Haematolymphoid Tumours
