Disease Pathogenesis
What is EBV+ PTLD?
- Patients who receive SOT or allogeneic HCT are at risk of developing PTLD due to immunosuppression 1–4
- EBV+ PTLD occurs as a result of deficient T cell surveillance and inadequate control of a primary EBV infection, or reactivation of a pre-existing, latent EBV infection, leading to unrestricted proliferation of transformed B cells1,3,5,6
EBV+ PTLD can be aggressive, life-threatening, and remains a challenging disease to manage2,4
Mechanism of Disease of EBV+ PTLD
-
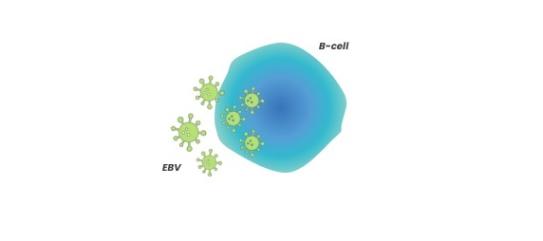
Primary infection occurs as the EBV invades host B cells, typically at a young age1
-
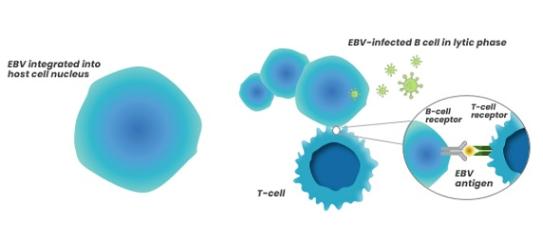
Cytotoxic T cells recognize infected B cells through expression of cell-surface viral proteins, however not all infected B cells are eliminated, causing a low-level latent infection that remains1,6
-
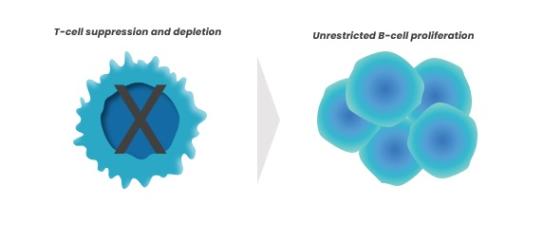
In immunocompromised patients (e.g., in the transplant setting), EBV cannot be controlled efficiently by EBV-specific cytotoxic T cells and may induce hyperproliferation of infected B cells, leading to EBV+ PTLD1,6
EBV+ PTLD as a complication arising from immunosuppression after SOT or HCT
SOT3
Immunosuppression
- Patients receive induction and maintenance therapy to prevent allograft rejection
- Induction therapy is given in the peri-transplantation period
- Maintenance therapy begins at transplantation and continues during the lifetime of the allograft
EBV Infection
- EBV-infected B cells proliferate due to impaired T-cell immunity; extended B-cell lifespan allows for genetic aberrations
A decrease in T-cell immune surveillance is a major contributing factor in the development of EBV+ PTLD
HCT2
Immunosuppression
- Patients receive conditioning regimens and immunosuppressive agents
- The pre-transplant conditioning regimen is given prior to donor stem cell infusion
- Immunosuppressive agents are given after transplant
EBV Infection
- EBV-infected B cells proliferate due to impaired T-cell immunity; extended B-cell lifespan allows for genetic aberrations
The combination of T-cell dysfunction plus cytokine-induced inflammation is a major risk factor for EBV+PTLD
Comparison of PTLD after HCT versus after SOT
|
|
SOT |
HCT |
|---|---|---|
|
Typical cell of origin |
Recipient origin2 |
Donor origin2 |
|
Incidence* |
1–33%2 |
0.8–4%2 |
|
EBV-associated PTLD |
~80%1 |
~100%8 |
*After SOT, incidence rates of PTLD may vary based on the type of organ transplanted, the recipient’s pre-transplant EBV antibody status, and the age of the recipient1; after HCT, incidence rates of PTLD may vary based on patient characteristics, stem cell source, degree of HLA mismatch, and conditioning regimen2
References
Abbreviations
- BCL6 B-cell lymphoma 6
- c-MYC cellular MYC
- EBV(+) Epstein–Barr virus (positive)
- GvHD graft-versus-host disease
- HCT haematopoietic cell transplantation
- HLA human leukocyte antigen
- IL-6 interleukin
- KMT2D lysine methyltransferase 2D
- mTOR mammalian target of rapamycin
- p53 tumour protein 53
- PTLD post-transplant lymphoproliferative disorder
- SOT solid organ transplantation
- SPEN Spen Family Transcriptional Repressor
- TET2 ten-eleven translocation 2
- TNF-α tumour necrosis factor-alpha
