Patient Management and Treatment Outcomes
Management of EBV+ PTLD and considerations for patient care
A core multidisciplinary team (MDT) should create a management plan for patient care1
Consider the patient's
- General health
- Degree of immunosuppression
- Histological subtype
- Stage of lymphoproliferative disorder
- Available therapy modalities
Reduction in immunosuppression
1L therapy
Maintained throughout treatment in some patients
Often in combination with rituximab
Rituximab ± chemotherapy
For patients with no response or disease progression
T-cell immunotherapy
For patients with R/R EBV+ PTLD or those for whom chemotherapy is inappropriate
SOT Key treatment outcomes
RIS
Key treatment outcomes: RIS
In a prospective, multicentre, Phase 2 study of 16 eligible adult SOT recipients with PTLD, RIS resulted in 1 out of 16 PRs (6%) and no CRs, supporting the addition of rituximab to RIS as initial therapy11
|
Patient characteristics11 |
||||
|---|---|---|---|---|
|
Median patient age was 47 years; 75% of patients had late PTLD, and |
||||
|
|
Management of immunosuppressives |
Length of time |
Response, |
Toxicity |
|
Group 1: Clinically urgent disease |
CsA/tacrolimus: reduced to 25% |
Immunosuppressives were maintained at this level for a maximum of 10 days |
CR: 0 (0%) |
Rejections: 6/16 (38%) |
|
Group 2: Clinically not urgent |
CsA/tacrolimus: reduced to 50% |
Immunosuppressives were maintained at this level for a maximum of 14 days |
||
Rituximab monotherapy
Key treatment outcomes: Rituximab monotherapy Overview
Prospective, multicentre, Phase 2 trials have investigated the efficacy and safety of rituximab in adults with PTLD after SOT who did not respond to prior RIS, demonstrating that treatment with rituximab was both effective and well-tolerated12–14
|
|
N |
Response, |
Infections |
Rituximab- |
Median OS |
|---|---|---|---|---|---|
|
Oertel, et al. (2005)12 |
17 |
CR: 9 (53%) |
None |
None |
37.0 |
|
Choquet, et al. (2006)13 |
43 |
CR: 12 (28%)*
|
None |
None |
14.9 |
|
González Barca, et al. (2007)14,† |
38 |
CR: 23 (61%) |
None |
None |
42.0 |
*Day 80 response: nine patients with CR; three patients with complete unconfirmed response.
†In case of partial remission, four additional doses of rituximab were administered.
Chemotherapy +/- rituximab
Key treatment outcomes: Chemotherapy overview
Retrospective studies have evaluated the clinical outcomes of CHOP-based chemotherapy in adults with PTLD after SOT, revealing high rates of toxicity16–20
|
Group |
Chemotherapy regimen (n) |
CR, % (n/n) |
Treatment-related mortality, |
|---|---|---|---|
|
Mamzer-Bruneel, et al. (2000)16 |
CHOP (8), CHOP+RT (2) |
60 (6/10) |
60 (6/10*) |
|
Norin, et al. (2004)17 |
CHOP (6), CHOP + MTX (2) |
50 (6/12) |
17 (2/12) |
|
Elstrom, et al. (2006)18 |
CHOP (10), R-CHOP (9), |
57 (13/23) |
26 (6/23) |
|
Fohrer, et al. (2006)19 |
ACVBP (32), |
67 (22/33) |
9 (3/33) |
|
Choquet, et al. (2007)20 |
CHOP (26) |
50 (13/26) |
31 (8/26) |
*All deaths regardless of causality; †CHOP + dexamethasone, ifosfamide, MTX, cytarabine, etoposide, intrathecal methotrexate, cytarabine, prednisolone; 2 patients received chemotherapy other than CHOP; ‡CEP: CTX, etoposide, prednisone; ESHAP: etoposide, methylprednisolone, cytarabine, cisplatin; ProMACECytaBOM: mechlorethamine, doxorubicin, cyclophosphamide, etoposide, vincristine, prednisone, procarbazine, methotrexate, cytarabine, bleomycin; R/C: rituximab, cyclophosphamide.
Sequential treatment (rituximab followed by chemotherapy)
Key treatment outcomes: PTLD-1 ST study – ST with rituximab then CHOP
In a prospective, open-label, Phase 2 multicentre study of patients with CD20+ PTLD who failed to respond to initial RIS, 90% of patients (53/59) treated with rituximab (once weekly for 4 weeks) followed by CHOP (four 21-day cycles) achieved a CR or PR response with completion of ST15
Treatment-related mortality was lower than previously reported by retrospective studies for 1L CHOP in PTLD15
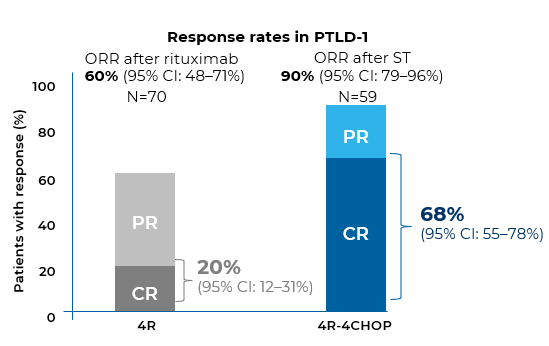
CHOP-associated treatment-related mortality:
10.6%
(7/66)
5 out of 7 CHOP-related deaths (71%) occured in non-responders to rituximab
Key treatment outcomes: PTLD-1 ST study
Median OS data supported that 1L ST was efficacious15

- ITT (N=70)
Median OS: 6.6 years
OS at 3 years: 61%
OS at 5 years: 55% - Analysis per protocol (N=63)
Median OS: not reached
OS at 3 years: 65%
OS at 5 years: 57%
Used with permission of Elsevier Ltd., from Sequential treatment with rituximab followed by CHOP chemotherapy in adult B-cell post-transplant lymphoproliferative disorder (PTLD): the prospective international multicentre phase 2 PTLD-1 trial. Trappe R., Oertel S., Leblond V., et al., Lancet Oncol. Vol. 34, 196–206 (2012); permission conveyed through Copyright Clearance Center, Inc.
Median TTP was 77.4 months (95% CI: 10.8–148.8): 69% (95% CI: 57–80%) of patients were progression-free at 3 years and 66% of patients (95% CI: 54–78) at 5 years15
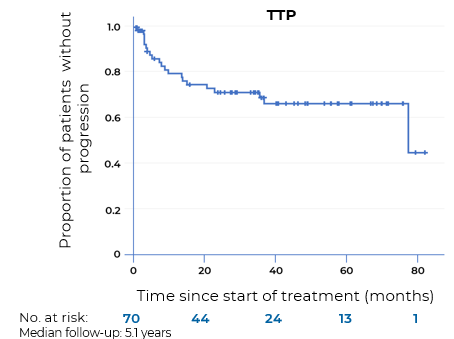
- The median TTP of patients treated according to the protocol (N=63) was not yet reached (>86.9 months) at the study cutoff dcate
without PD at 3 years: 70%
without PD at 5 years: 66% - Of the 40 patients who achieved CR, 8 patients (20%) relapsed
Used with permission of Elsevier Ltd., from Sequential treatment with rituximab followed by CHOP chemotherapy in adult B-cell post-transplant lymphoproliferative disorder (PTLD): the prospective international multicentre phase 2 PTLD-1 trial. Trappe R., Oertel S., Leblond V., et al., Lancet Oncol. Vol. 34, 196–206 (2012); permission conveyed through Copyright Clearance Center, Inc.
Key treatment outcomes: PTLD-1 RSST study
The subsequent prospective Phase 2 PTLD-1 RSST trial tested a response-adapted treatment strategy based on patient response to rituximab monotherapy induction and established that rituximab consolidation for patients in CR after rituximab induction was safe and effective21
Proportion of patients without progression at 3 years by treatment cohort
RSST (8R)
n=37
95% CI: 76-100%
ST (4R-4CHOP)
n=14
95% CI: 44-95%
At 3 years, improved TTP outcomes were observed in the low-risk rituximab consolidation group* compared to the patients in the PTLD-1 ST cohort who had reached CR with rituximab induction and continued ST with CHOP chemotherapy
*patients with CR at interim staging in the low-risk group (RSST cohort)
Key treatment outcomes: PTLD-2 study
A prospective, multicentre, Phase 2 trial tested a modified RSST treatment. In addition to patients who achieved a CR after rituximab induction, rituximab maintenance monotherapy was also given to those in PR and who were considered low risk with <3 IPI risk factors. Similar TTP, OS, and PFS were observed compared to the PTLD-1 ST and RSST trials22
TTP and OS in the ITT population
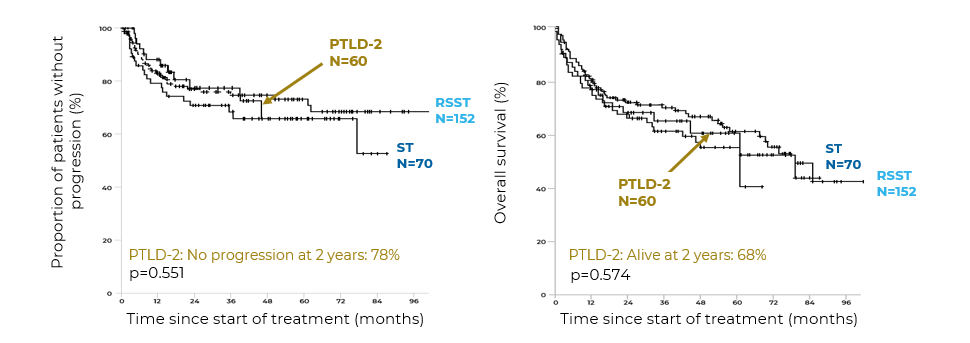
Used with permission of Springer Nature BV, from Modified risk-stratified sequential treatment (subcutaneous rituximab with or without chemotherapy) in B-cell Post-transplant lymphoproliferative disorder (PTLD) after Solid organ transplantation (SOT): the prospective multicentre phase II PTLD-2 trial. Zimmermann H., Koenecke C., Dreyling MH., et al., Leukemia. Vol. 36, 2468–2478 (2022); permission conveyed through Copyright Clearance Center, Inc.
Pooled analysis of low-risk groups in the PTLD-1-RSST and PTLD-2 trials (N=58)
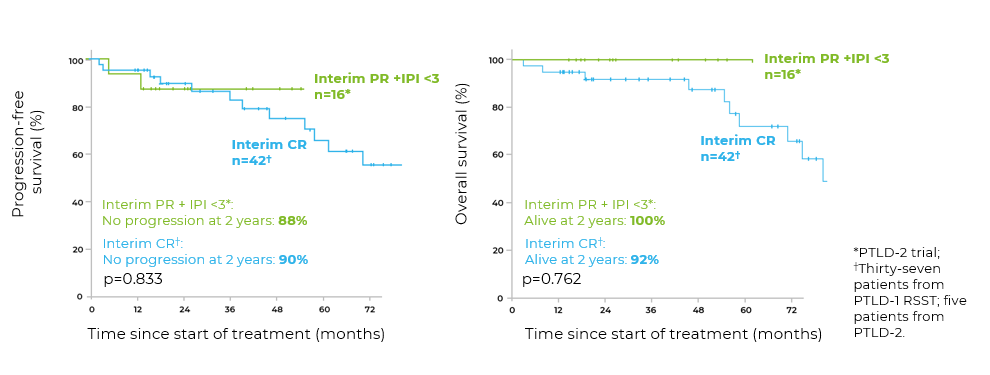
Adapted in color from Zimmermann H, et al. Leukemia. 2022. https://creativecommons.org/licenses/by/4.0/.
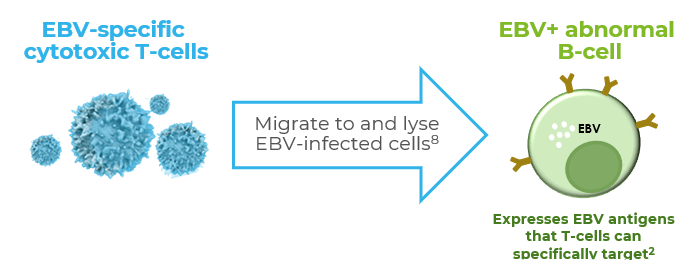
EBV-specific CTL immunotherapy
Key treatment outcomes: EBV-specific CTL immunotherapy
Ongoing unmet clinical need
Survival among patients whose disease is refractory to 1L treatment is low and prognosis remains poor23
SOT recipients

4.1
MONTHS
n=86
MEDIAN OS
In a retrospective chart review of patients with EBV+ PTLD following SOT who received rituximab alone or rituximab plus chemotherapy as initial therapy, median OS was 4.1 months from the earliest date when patients developed R/R disease23

Effective treatment options are needed to fulfil the unmet medical need for patients who fail available therapies
Experience with third-party EBV-CTLs suggest that EBV-CTLs should be explored as therapy for rituximab-refractory, EBV-positive PTLD emerging after SOT24
Adoptive T-cell therapy in the EBV+ PTLD treatment setting after SOT: third party EBV-CTLs
|
Study |
Method of selection |
Prior therapy |
n |
HLA |
Response (CR+PR) |
|---|---|---|---|---|---|
|
Mahadeo, et al.25* |
EBV-BLCL-stimulated EBV CTLs |
Rituximab ± CT |
29 |
≥2 |
52 (15/29) |
|
Bonifacius, et al.26* |
EBV-BLCL-stimulated T-cell line |
CT ± rituximab |
5 |
≥3/6 |
80 (4/5) |
|
Prockop, et al.27* |
EBV-BLCL-sensitised T-cell line |
Rituximab ± CT/RT |
13 |
≥2/10 |
54 (7/13) |
|
Chiou, et al.28 |
EBV-BLCL-stimulated EBV CTLs |
RIS ± rituximab/IG/antiviral |
10 |
2-5/6; 5-7/10 |
80 (8/10) |
|
Gallot, et al.29* |
EBV-BLCL-stimulated EBV CTLs |
CT ± rituximab/RT |
3 |
≥2 |
33 (1/3) |
|
Vickers, et al.30* |
EBV-LCL-stimulated EBV CTLs |
NA |
5 |
3-9/10 |
100 (5/5) |
|
Gandhi, et al.31 |
EBV-LCL-sensitised EBV CTLs |
RIS, rituximab ± CT |
3 |
≥3/6 |
66 (2/3) |
|
Haque, et al.32* |
EBV-LCL-stimulated EBV CTLs |
RIS, rituximab ± other |
31 |
2-5/6 |
61 (19/31) |
|
Sun, et al.33* |
EBV-BLCL-stimulated EBV CTLs |
RT; Rituximab + CT |
1; 1 |
4/6; 6/6 |
100(1/1);100(1/1) |
*These studies also investigated third-party EBV-CTLs in patients who received HCT.
Key Takeaways
Key takeaways for the management of EBV+ PTLD after SOT
There are a range of
therapy options for treating EBV+ PTLD
A core multidisciplinary team is important for creating an effective management plan
Patients not responding to 1L therapy face poor outcomes (4.1m mOS post-SOT in R/R EBV+ PTLD patients23) and limited alternative treatments
Adoptive
T-cell immunotherapy is a novel treatment option for patients with EBV+ PTLD who fail initial therapy
HCT Key treatment outcomes
RIS + rituximab, chemotherapy, DLI
Key treatment outcomes: Retrospective analyses
Variable response and limited 1- and 2-year survival rates with 1L rituximab indicate a need for improved treatment options for patients with EBV+ PTLD after HCT
of patients achieve CR to 1L ritixumab, highlighting the variability in response24,34
Reported 1- and 2-year survival rates in patients with EBV+ PTLD, the majority of whom (47/51) received rituximab as 1L treatment34
About 50% of EBV+ PTLD cases relapse or are refractory to initial treatment with rituximab-containing regimens,35 and only a few studies have reported treatment outcomes in patients with rituximab-refractory EBV+ PTLD:
|
|
n/N* |
Type of treatment (n) |
Response rate |
Relapses |
|---|---|---|---|---|
|
Hou (2009)36 |
1/12† |
RIS + rituximab (1); |
CR: 1/1 (RIS + rituximab) |
- |
|
Fox (2014)34 |
3/62‡ |
CHOP (5); |
CR: 0/5, PR: 0/5 (CHOP) |
None |
|
Socié (2024)35 |
4/81‡ |
Chemotherapy-containing regimen (32); |
Durable response of >6 months: 4/36 |
2/4 |
*n: the total number of patients who achieved a CR; N: the number of patients constituting the study population.
†One patient achieved CR after 1L therapy.
‡Patients who achieved a CR (or durable response) after next-line therapy.
EBV-specific CTLs
Key treatment outcomes: Ongoing unmet clinical need
Survival among patients whose disease is refractory to 1L treatment is low and prognosis remains poor35
HCT recipients

0.7
MONTHS
n=81
MEDIAN OS
In a retrospective chart review of allogeneic HCT recipients who failed rituximab alone or rituximab plus chemotherapy, the median OS was 0.7 months from the time of initial treatment failure leading to R/R EBV+ PTLD35

Effective treatment options are needed to fulfil the unmet medical need for patients who fail available therapies
Treatment with EBV-specific CTLs generated from primary HCT donors was found to be effective in the treatment of EBV+ PTLD; EBV-specific CTLs have been effective in >80% of patients treated for PTLD24
Adoptive T-cell therapy in the EBV+ PTLD treatment setting after HCT: donor-derived EBV-CTLs24
|
Study |
Method of selection |
n |
Response (CR+PR) |
Acute GvHD |
|---|---|---|---|---|
|
Rooney, et al. |
EBV-LCL sensitised |
13 |
85 (11/13) |
8% |
|
Lucas, et al. |
EBV-LCL sensitised |
1 |
100 (1/1) |
100% |
|
Imashuku, et al. |
EBV-LCL sensitised |
1 |
0 (0/1) |
0% |
|
Gottschalk, et al. |
EBV-LCL sensitised |
1 |
0 (0/1) |
Not documented |
|
Comoli, et al. |
EBV-LCL sensitised |
5 |
100 (5/5) |
0% |
|
Moosman, et al. |
Peptide stimulation IFN𝛾 capture |
6 |
50 (3/6) |
0% |
|
Doubrovina, et al. |
EBV-LCL sensitised |
19 |
68 (13/19) |
0% |
|
Icheva, et al. |
EBNA-1 peptide stimulation IFN𝛾 capture |
8 |
75 (6/8) |
13% |
|
Gerdemann, et al. |
DC nucleofection viral plasmids |
1 |
100 (1/1) |
100% |
|
Papadopoulou, et al. |
Peptide-stimulated |
1 |
100 (1/1) |
0% |
Adapted from Shahid S, et al. Cancer Drug Resist. 2021. https://creativecommons.org/licenses/by/4.0/.
Adoptive T-cell therapy in the EBV+ PTLD treatment setting after HCT: third-party EBV-CTLs
|
Study |
Method of selection |
Prior therapy |
n |
HLA |
Response (CR+PR)% (n/n) |
|---|---|---|---|---|---|
|
Mahadeo, et al.25* |
EBV-BLCL-stimulated EBV CTLs |
Rituximab |
14 |
≥2 |
50 (7/14) |
|
Bonifacius, et al.26* |
EBV-BLCL-stimulated T-cell line |
Rituximab ± CT |
14 |
≥3/6 |
60 (6/10)† |
|
Prockop, et al.27* |
EBV-BLCL-sensitised T-cell line |
Rituximab ± CT/RT |
33 |
≥2/10 |
67 (22/33) |
|
Tzannou, et al.37 |
Peptide-stimulated CTLs |
Antiviral therapy; None |
1; 1 |
3/8; 5/8 |
100 (1/1); |
|
Naik, et al.38 |
EBV-LCL- and multi-virus-sensitised CTLs |
None/other or rituximab ± other |
12 |
varies |
75 (9/12) |
|
Gallot, et al.29* |
EBV-LCL-stimulated EBV CTLs |
CT ± rituximab |
6 |
≥2 |
50 (3/6) |
|
Vickers, et al.30* |
EBV-BLCL-stimulated EBV-CTLs |
NA |
6 |
3-9/10 |
67 (4/6) |
|
Leen, et al.39 |
Transduced multi-virus CTLs |
Rituximab |
9 |
1-3/6 |
67 (6/9) |
|
Barker, et al.40 |
EBV-BLCL-stimulated T-cell line |
Rituximab ± RT |
2 |
5/6, 4/6 |
100 (2/2) |
|
Haque, et al.32* |
EBV-LCL-stimulated EBV CTLs |
RIS ± rituximab ± other |
2 |
2–5/6 |
100 (2/2) |
*These studies also investigated third-party EBV-CTLs in patients who received SOT. †Outcome data was available for 10 patients.
Key takeaways
Key takeaways for the management of EBV+ PTLD after HCT
The management of EBV+ PTLD is complex, with limited treatment options for patients who are relapsed/refractory to initial therapy
Patients not responding to 1L therapy face poor outcomes (0.7m mOS post-HCT in R/R EBV+ PTLD patients35)
Adoptive
T-cell immunotherapy serves as an alternative treatment option to help improve outcomes for patients with R/R EBV+ PTLD
References
- Shah N, et al. Br J Haematol. 2021;193(4):727–740.
- Nijland ML, et al. Transplant Direct. 2015;2(1):e48.
- Yamada M, et al. J Pediatric Infect Dis Soc. 2024;13(Suppl 1):S31–S38.
- Lindsay J, et al. Curr Opin Infect Dis. 2021;34(6):635–645.
- Sureda A, et al. The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies. 8th edition. Cham (CH): Springer; 2024.
- Styczynski J, et al. Haematologica. 2016;101(7):803–811.
- Dierickx D, Habermann TM. N Engl J Med. 2018;378(6):549–562.
- Styczynski J, et al. Anticancer Res. 2022;42:5181-5186.
- Clerico M, et al. J Clin Med. 2022;11(24):7542.
- Wistinghausen B, et al. Ann Lymphoma. 2022;6:5.
- Swinnen LJ, et al. Transplantation. 2008;86(2):215–222.
- Oertel SH, et al. Am J Transplant. 2005;5(12):2901–2906.
- Choquet S, et al. Blood. 2006;107(8):3053–3057.
- González-Barca E, et al. Haematologica. 2007;92(11):1489–1494.
- Trappe R, et al. Lancet Oncol. 2012;13(2):196–206.
- Mamzer-Bruneel MF, et al. J Clin Oncol. 2000;18(21):3622.
- Norin S, et al. Med Oncol. 2004;21(3):273–284.
- Elstrom RL, et al. Am J Transplant. 2006;6(3):569–576.
- Fohrer C, et al. Br J Haematol. 2006;134(6):602–612.
- Choquet S, et al. Haematologica. 2007;92(2):273–274.
- Trappe R, et al. J Clin Oncol. 2017;35:536–543.
- Zimmermann H, et al. Leukemia. 2022;36(10):2468–2478.
- Dharnidharka V, et al. Hemasphere. 2022; 6(Suppl): 997–998.
- Shahid S, Prockop SE. Cancer Drug Resist. 2021;4(3):646–664.
- Mahadeo KM, et al. Lancet Oncol. 2024;25(3):376-387.
- Bonifacius A, et al. J Clin Invest. 2023;133(12):e163548.
- Prockup S, et al. J Clin Invest. 2020;130(2):733–747.
- Chiou FK, et al. Pediatr Transplant. 2018;22(2).
- Gallot G, et al. J Immunother. 2014;37(3):170-9.
- Vickers MA, et al. Br J Haematol. 2014;167(3):402-10.
- Gandhi MK, et al. Am J Transplant. 2007;7(5):1293-9.
- Haque T, et al. Blood. 2007;110(4):1123-31.
- Sun Q, et al. Br J Haematol. 2002;118(3):799-808.
- Fox CP, et al. Bone Marrow Transplant. 2014;49(2):280–286.
- Socié G, et al. Bone Marrow Transplant. 2024;59(1):52–58.
- Hou HA, et al. Bone Marrow Transplant. 2009;43(4):315–321.
- Tzannou I. J Clin Oncol. 2017;35(31):3547-3557.
- Naik S, et al. J Allergy Clin Immunol. 2016; 137(5): 1498–1505.e1.
- Leen AM, et al. Blood. 2013;121(26):5113-23.
- Barker JN, et al. Blood. 2010;116(23):5045-9.
Abbreviations
- 1L first-line
- 4CHOP four cycles of CHOP
- 4R four courses of rituximab
- 8R eight courses of rituximab
- ACVBP doxorubicin, cyclophosphamide, vindesine, bleomycin, and prednisone
- AZA azathioprine
- BLCL EBV-transformed B-lymphoblastoid cell
- BOR best overall response
- CD20 cluster of differentiation 20
- CEP cyclophosphamide, etoposide, prednisone
- CHOP cyclophosphamide, doxorubicin hydrochloride (hydroxydaunorubicin), vincristine sulfate (Oncovin), and prednisone
- CI confidence interval
- CR complete response
- CsA cyclosporine
- CTLs cytotoxic T lymphocytes
- CTX cyclophosphamide
- DC dendritic cell
- DLBCL diffuse large B-cell lymphoma
- DLI donor lymphocyte infusion
- EBNA-1 Epstein-Barr nuclear antigen 1
- EBV(+) Epstein–Barr virus (positive)
- EMA European Medicines Agency
- ESHAP etoposide, methylprednisolone, cytarabine, cisplatin
- GvHD graft-versus-host disease
- HCT haematopoietic cell transplantation
- HLA human leukocyte antigen
- IFNγ interferon‐gamma
- IPI International Prognostic Index
- IS immunosuppression
- ITT intent-to-treat
- IV intravenous
- LCL lymphoblastoid cell lines
- LDH lactate dehydrogenase
- MDT multidisciplinary team
- MTX methotrexate
- NA not available
- ORR overall response rate
- OS overall survival
- PD disease progression
- PFS progression-free survival
- PR partial response